UBC zoologist solves the 100-year-old mystery of the floating phantom midge
January 26, 2022
January 26, 2022

In spring 2018, Dr. Philip Matthews spent a typical afternoon capturing dragonflies in the University of British Columbia’s (UBC) experimental ponds. Little did the zoologist know he was about to embark on a journey to solve a century-old entomological mystery involving a much smaller, but equally intriguing, insect. As he worked in the ponds, larvae floating in rainwater in a nearby cattle tank caught his eye.
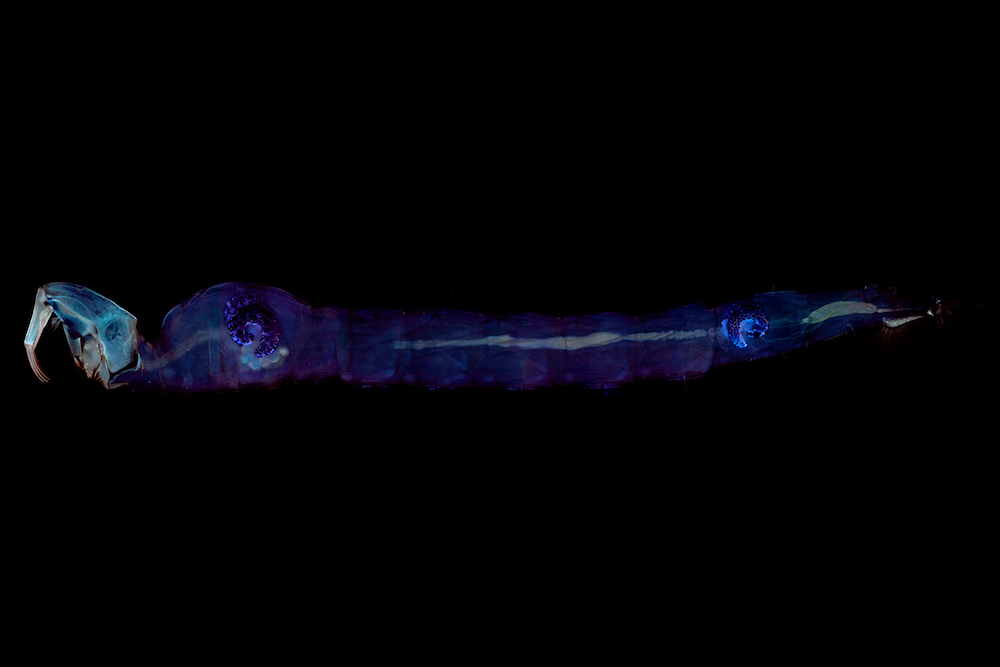
The insects were the freshwater aquatic larvae of the Chaoborus midge, also called the ‘phantom midge’ due to its near transparency. The transparency makes the larvae resemble tiny ghosts as they move through lakes, ponds and puddles.
“These bizarre insects were floating neutrally buoyant in the water, which is something you just don't see insects doing,” said Dr. Matthews. “Some insects can become neutrally buoyant for a short time during a dive, but Chaoborus larvae are the only insects close to being neutrally buoyant.”
Some fish regulate their buoyancy by inflating a swim bladder with oxygen unloaded from the hemoglobin in their blood. In 1911, Nobel laureate August Krogh discovered Chaoborus larvae use a completely different mechanism, regulating their buoyancy using two pairs of internal air-filled sacs. But he never figured out how the insects adjusted the volume of their sacs without having blood or hemoglobin like vertebrates do.
Back in the lab after his coffee, Dr. Matthews mounted the air-sacs of the larvae from the cattle tank on a microscope that just happened to have ultraviolet light illuminating the microscope’s stage. The air-sacs started glowing blue.
The blue fluorescence was due to resilin—an almost perfect rubber found in parts of insects where elasticity is key, as in the elastic energy that powers a flea’s incredible jump.
“The weird thing about resilin is that not only is it really elastic. It will swell if you make it alkaline and contract if you make it acidic.”
With PhD student Evan McKenzie driving experimental investigations, the researchers discovered that the insect doesn’t secrete gas into their air-sacs to make them expand. Instead, they change the pH level of the air-sac wall, the bands of resilin within the air-sac wall swell or contract in response, and the volume of the sac adjusts.
“This is a really bizarre adaptation that we didn't go looking for,” says Dr. Matthews. “We were just trying to figure out how they can float in water without sinking!”
The Chaoborus air-sacs function as mechanochemical engines, converting changes in chemical potential energy into mechanical work.
The findings were published this week in Current Biology.
Chaoborus uses its ability to change buoyancy to travel vertical distances very cheaply in terms of energy expended. They don’t have to swim at all to move through the water. It also allows the larvae to occupy the mid-water zone, giving them a whole region of the aquatic environment that most insects can’t use well.
Of course, moving around in the mid-water zone means the larvae are prey for fish. Midge larvae that live in lakes have to hide on the bottom in sediment during the day so they aren’t eaten. Then at night, when they are less visible to fish, they rise to the surface to feed on zooplankton.
The larvae of one species, C.edulis, live in Lake Malawi in east Africa which is 700 metres deep at some points. That’s far too deep for the one centimetre-long larvae to reach the lakebed after a night of feeding. But the lake is stratified with a hypoxic layer around 200 meters deep where the fish cannot survive and the midge larvae routinely descend 250 metres below the surface where they are safe from predation.
“The pond species tend to live in ponds that don't have fish,” says Dr. Matthews.
We honour xwməθkwəy̓ əm (Musqueam) on whose ancestral, unceded territory UBC Vancouver is situated. UBC Science is committed to building meaningful relationships with Indigenous peoples so we can advance Reconciliation and ensure traditional ways of knowing enrich our teaching and research.
Learn more: Musqueam First Nation